VALCHLOR® (mechlorethamine) gel is an alkylating drug indicated for the topical treatment of Stage IA and IB mycosis fungoides–type cutaneous T-cell lymphoma (MF-CTCL) in patients who have received prior skin-directed therapy
The PROVe study:
US real-world experience with mechlorethamine gel in combination with other therapies
ADDITIONAL DATA NOT CONTAINED IN THE USPI
The folowing information is from a prospective observational analysis providing real-world data on mechlorethamine gel (VALCHLOR) when used concomitantly with other therapies for patients with Stage IA/IB MF-CTCL who have received prior skin-directed therapy.1
The PROVe data provides supplementary information about response rates, safety, and tolerability in patients using VALCHLOR with concomitant therapies. This information is not contained in the FDA-approved label. Use of VALCHLOR with concomitant therapies has not been evaluated by FDA.
These data is being provided as background information to help healthcare professionals when making assessments to improve patient care. The PROVe data should not be interpreted as demonstrating better efficacy than what has been evaluated and approved in the product labeling.
Real world use of mechlorethamine gel evaluated with concomitant therapies
Study design and population
The PROVe (a PROspective, observational study assessing outcomes, adverse events [AEs], treatment patterns, and quality of life [QOL] in patients diagnosed with mycosis fungoides–type cutaneous T-cell lymphoma (MF-CTCL) and treated with VALCHLOR and other therapies) study was designed to examine the real-world use of chlormethine gel in adult patients with MF-CTCL in routine clinical practice across the US.1
PROVe enrolled 298 patients. Clinical response data was available for portions of the total enrolled patient population as scored by investigators. The analyses were pre-specified, descriptive, and not statistically powered. Therefore, this information should be cautiously interpreted.1
Multicenter, prospective, observational, US-based, noninterventional study1
- All consecutive patients ≥18 years with a diagnosis of MF-CTCL being treated with mechlorethamine gel at each of the 46 participating centers could enroll
- Mean age, 61.7 years; 60.1% male; mean duration of MF-CTCL, 5.5 years
- Patients (N=298) were included regardless of disease stage or previous and concomitant therapy (received before or at enrollment) and were followed for up to 2 years
- 62.4% of patients had stage IA-IIA disease at enrollment; 8.4% had IIB-IV disease, and current staging was unavailable for 29.2% (VALCHLOR is only indicated for Stage IA and IB MF-CTCL2)
- No exclusion criteria were applied
-
4 patient groups based on a predominant concomitant MF-CTCL therapy at enrollment
- Mechlorethamine gel + topical corticosteroids + other (VCO; n=185)
- Mechlorethamine gel + phototherapy + other (VPO; n=76)
- Mechlorethamine gel + oral bexarotene + other (VOB; n=47)
- Mechlorethamine gel + any other treatment (VO; n=298)
- Flexible dosing was permitted in the PROVe study, therefore some patients were instructed by investigators to use mechlorethamine gel less than once daily
Primary and other specific treatments used in ≥10% of patients by group at enrollment
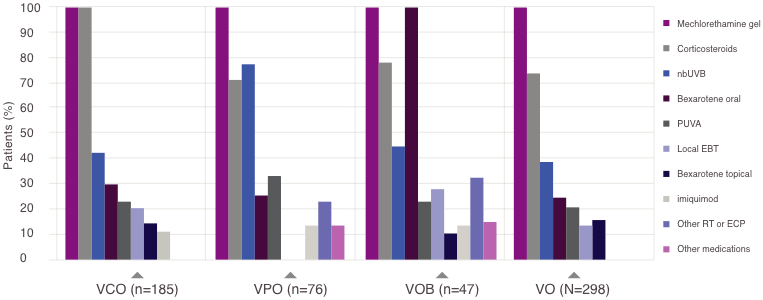
EBT=electron-beam therapy; ECP=extracorporeal photopheresis; nbUVB=narrow-band ultraviolet B; PUVA=psoralen and ultraviolent A; RT=radiotherapy.
-
Primary efficacy endpoint: proportion of patients in the VCO group with stage IA-IB disease and evaluable response; response was defined as ≥50% reduction in percentage of affected body surface area (%BSA) from baseline at 12 months1
- Secondary efficacy endpoints: %BSA response rates at 12 months in the VPO, VOB, and VO groups; by-time analysis of the %BSA response rates at months 1, 3, 6, 9, 12, 15, 18, 21 and 24 (±45 days)
- Overall response rate (ORR): the proportion of patients with a ≥50% reduction from baseline in %BSA for 2 consecutive visit
Study limitations
As no specific assessments were mandated, there was a limited number of patients who had both pre-enrollment and postbaseline %BSA data available for analysis. In addition, the clinical responses reported herein likely also reflect the use of concomitant therapies. Treatment schedules and frequency of mechlorethamine gel use varied, and patients could also have multiple dosing regimens over time, which can complicate data analysis but is representative of mechlorethamine gel use in daily clinical practice.
PROVe study findings
Duration of mechlorethamine gel treatment and use of other therapies
- Median duration of mechlorethamine gel treatment—24 months for newly initiated patients; 32 months for patients using mechlorethamine gel for ≥3 months at enrollment1
- Concomitant therapy on study—77.9% had other skin-directed therapies; 30.2% had systemic therapies1
%BSA response rates
- ORR at 12 months in patients with stage IA-IB disease: 44.4% (24/54) VCO group; 45.1% (37/82) VO group1
- By-time analysis: designed to collect information about response over time, providing complementary data to the ORR to help visualize the changing response rates during treatment1
By-time analysis of the %BSA response data
Clinical responses occurred as early as 1 month after treatment, with peak response occurring at 18 months for patients with stage IA-IB disease in the VO group (66.7%)1
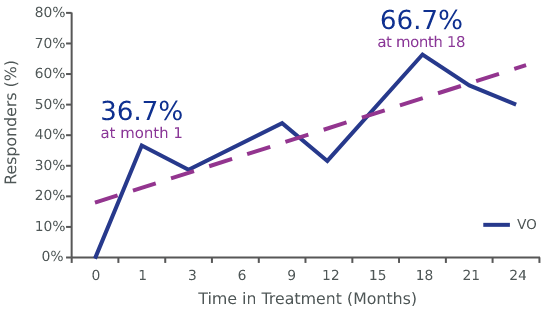
%BSA was analyzed as a surrogate measurement of response. The %BSA did not take severity of lesions into account and could result in significant underestimation of the response data.
Safety
Safety evaluation: documentation of AEs and serious AEs at every visit and determining their relation to VALCHLOR1
- No treatment-related serious AEs or deaths were reported1
| Common Adverse Events (≥3.0%) | Mechlorethamine gel + Other (N=298) | |
|---|---|---|
| Overall | 125 (41.9%) | 83 patients (27.9%) experienced treatment-related AEs; the most common skin-related AEs were dermatitis, pruitus, skin irritation, and erythema1 |
| Dermatitis | 38 (12.8%) | |
| Pruritus | 29 (9.7%) | |
| Skin Irritation | 22 (7.4%) | |
| Erythema | 15 (5.0%) | |
| Skin burning sensation | 11 (3.7%) | |
| Rash | 10 (3.4%) |
